 
Developers and engineers get so immersed in the “design and refine” process that they sometimes forget about their obligation to document how they got from A to B. 
Yet, due to the nature of medical devices and their impact on personal health and safety, the entire product life cycle of a device must be carefully documented. 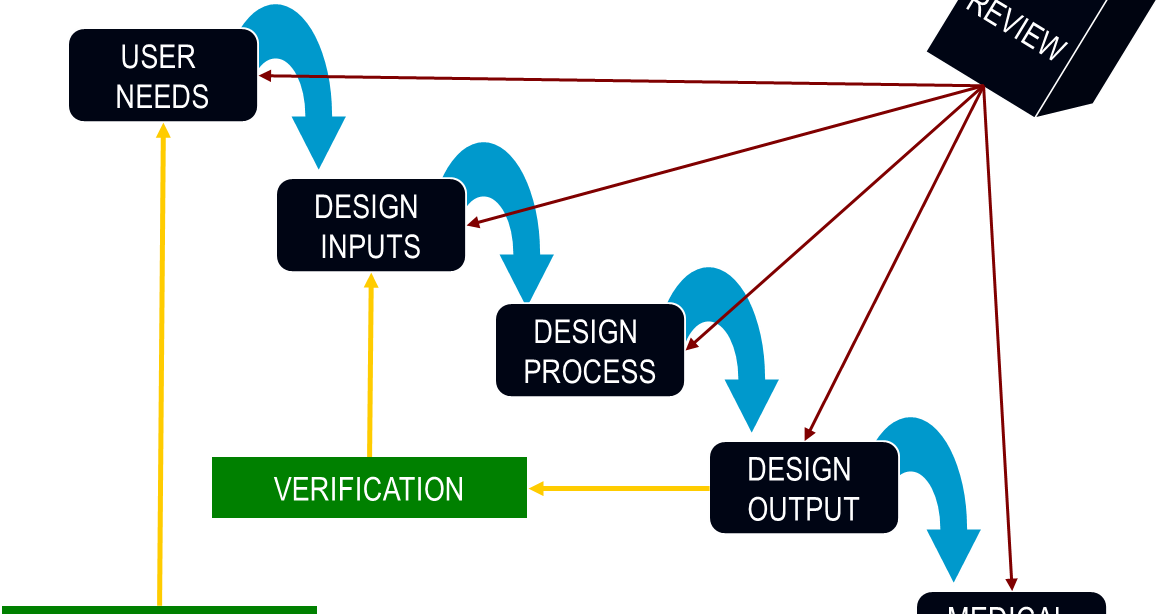
Medical devices, like all products and software, are constantly evolving. We’ve combined all three posts into one easy-to-read PDF, plus added some extras. In subsequent post we look at design inputs and outputs, and then DHR, DHF and DMRs. 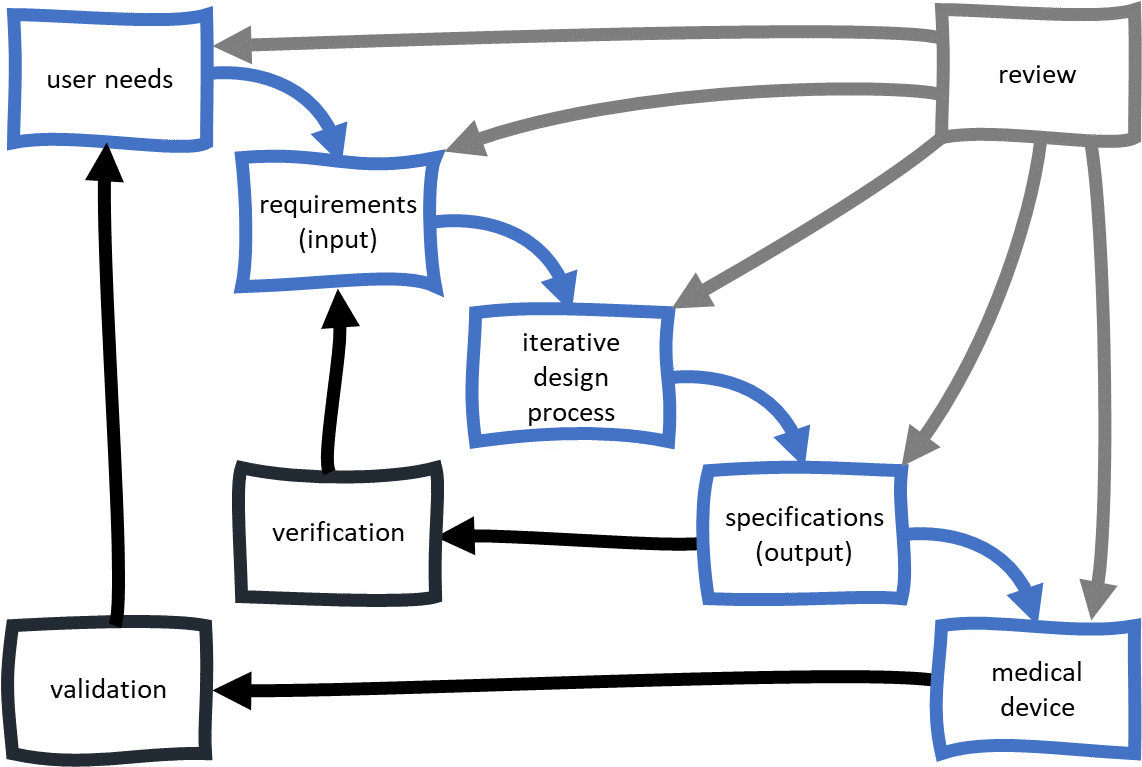
This is the first post in a 3-part blog series on medical device design control. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
